Market Overview:
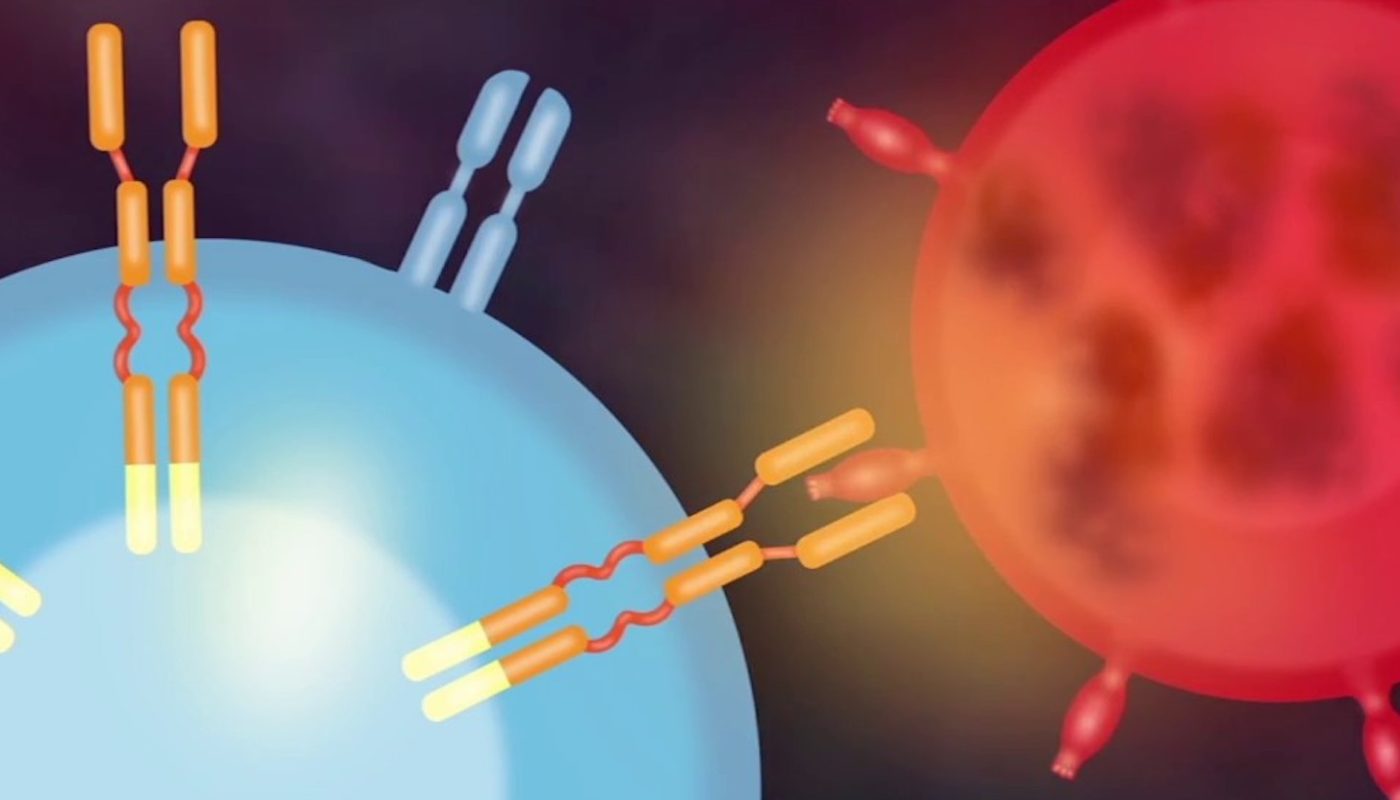
CAR T Cell Therapy is a revolutionary approach to cancer treatment that involves genetically modifying a patient’s own immune cells to recognize and attack cancer cells. This therapy has gained significant attention and traction in recent years due to its high success rates in treating certain types of blood cancers, such as leukemia and lymphoma.
CAR T Cell Therapy offers several advantages over traditional cancer treatments, including higher response rates, longer remission periods, and potentially lower risks of relapse. The need for effective and targeted treatments for cancer patients, especially those who have failed conventional therapies, has driven the adoption of CAR T Cell Therapy.
Market Key Trends:
One key trend in the CAR T Cell Therapy market is the increasing adoption of off-the-shelf CAR T Cell therapies. Traditional CAR T Cell therapies require the extraction and modification of a patient’s own immune cells, which can be time-consuming and costly. Off-the-shelf therapies, on the other hand, use pre-engineered CAR T cells from healthy donors, eliminating the need for individualized patient manufacturing.
For example, Fate Therapeutics is developing an off-the-shelf CAR T Cell therapy called FT819, which utilizes donor-derived CAR T cells and has shown promising results in preclinical studies. This approach has the potential to streamline the manufacturing process and improve accessibility and affordability of CAR T Cell Therapy.
Porter’s Analysis:
The threat of new entrants:
The entry barriers in the CAR T cell therapy market are high, mainly due to the complex manufacturing process, stringent regulatory requirements, and significant investment in research and development. This limits the threat of new entrants.
Bargaining power of buyers:
The bargaining power of buyers in the CAR T Cell Therapy market is relatively low due to the limited availability of alternative treatments and the critical nature of the therapy for patients.
Bargaining power of suppliers:
The bargaining power of suppliers in the CAR T Cell Therapy market is moderate, as there are several suppliers of raw materials and reagents required for the manufacturing process.
The threat of new substitutes:
Currently, CAR T Cell Therapy has limited substitutes for the treatment of certain types of blood cancers. However, the development of alternative immunotherapies or targeted therapies could pose a threat in the future.
Competitive rivalry:
The CAR T Cell Therapy market is highly competitive, with several key players vying for market share. The market is characterized by intense research and development activities, strategic collaborations, and mergers and acquisitions.
Key Takeaways:
The global CAR T Cell Therapy market is expected to witness high growth, exhibiting a CAGR of 20.9% over the forecast period, due to increasing demand for effective and targeted cancer treatments. The success of CAR T Cell Therapy in treating certain types of blood cancers has attracted significant attention and investment in the market.
In terms of regional analysis, North America is expected to dominate the CAR T Cell Therapy market, driven by well-established healthcare infrastructure, favorable reimbursement policies, and high adoption of innovative therapies. Asia Pacific is expected to be the fastest-growing region, attributed to the growing prevalence of cancer, increasing healthcare expenditure, and improving access to advanced treatments.
Key players operating in the global CAR T Cell Therapy market include Fate Therapeutics, Mustang Bio, Sorrento Therapeutics, Inc., Bluebird bio, Inc., Pfizer Inc., Gilead Sciences, Inc., Legend Biotech, Aurora Biopharma, CARsgen Therapeutics Co., Ltd, Novartis AG, Johnson & Johnson Services, Inc., and Bristol-Myers Squibb Company. These companies are actively engaged in research and development activities, clinical trials, and strategic collaborations to enhance their market presence and expand their product portfolios.