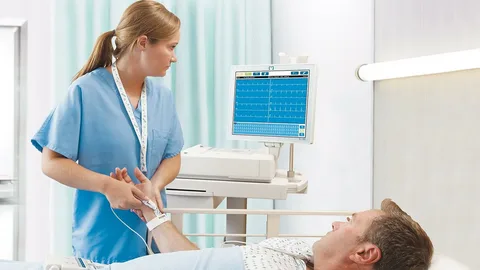
A research team from the MIT Department of Aeronautics and Astronautics (AeroAstro) Engineering Systems Lab and Synensys, a safety management contractor, has conducted a study to examine the ecosystem of diagnostic laboratory data and identify factors contributing to patient hazards in laboratory diagnostic tests. The team’s findings offer a holistic view of the complex network involved in diagnostic laboratory data, including doctors, lab technicians, device manufacturers, health information technology (HIT) providers, and government entities.
The study, led by AeroAstro Professor Nancy Leveson, along with Research Engineer John Thomas and graduate students Polly Harrington and Rodrigo Rose, submitted a report to the U.S. Food and Drug Administration (FDA) last fall. The FDA has prioritized improving the infrastructure of laboratory data and contracted the study through Synenisys.
Through interviews with more than 50 stakeholders, the research team found that the diagnostic laboratory data ecosystem is extensive but fragmented, with no clear understanding of how the entire system functions or the full extent of substandard treatment provided to patients. The MIT engineers discovered numerous hazards, including test results being sent to the wrong patients, incompatible technologies hindering information sharing between doctors and lab technicians, and specimens transported to the lab without proper temperature control.
To address these hazards, the research team identified six systemic factors contributing to patient safety risks in laboratory diagnostics. Four of these factors—decentralization, flawed communication and coordination, insufficient focus on safety-related regulations, and ambiguous or outdated standards—highlight the need for enhanced oversight and accountability. The remaining two factors—misperceived notions of risk and lack of integration of systems theory—call for a fundamental shift in perspective and operations.
The report emphasizes that blaming individual physicians for errors is unreasonable and unfair when the data they receive is assumed to be accurate. The medical community needs to adopt systems theory, a concept well-established in the aviation industry, to understand the real risk levels associated with laboratory data and HIT.
The aviation industry has successfully implemented systems theory to enhance safety. After World War II, when commercial aviation crashes were frequent, the industry and government collaborated to introduce controls and error reporting, leading to the near elimination of crashes in the U.S. Applying engineering principles, such as closed-loop controls, to healthcare could also improve patient safety. Closed-loop controls, which solicit feedback and allow for course corrections, are currently lacking in the healthcare system.
The research team, in collaboration with Synensys, will continue working with the FDA on their next study, which will investigate diagnostic screenings outside the laboratory, such as at a physician’s office or at home. The COVID-19 pandemic has significantly increased nonclinical lab testing, but systems are lacking to aggregate data and report findings to public health agencies.
By considering healthcare as an engineered system, the researchers believe that it can help solve entrenched problems and improve patient safety. The findings of this study provide valuable insights for healthcare stakeholders to address systemic factors and implement changes that will lead to safer practices and better patient outcomes.
Note:
1. Source: Coherent Market Insights, Public sources, Desk research
2. We have leveraged AI tools to mine information and compile it